Amorphous solid
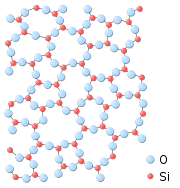
An "amorphous solid" is a solid in which there is no long-range order of the positions of the atoms. (Solids in which there is long-range atomic order are called crystallines or morphous). Most classes of solid materials can be found or prepared in an amorphous form. For instance, common window glass is an amorphous solid, many polymers (such as polystyrene) are amorphous, and even junkfoods such as cotton candy are amorphous solids.
In principle, given a sufficiently high cooling rate, any liquid can be made into an amorphous solid. Cooling reduces molecular mobility. If the cooling rate is faster than the rate at which molecules can organize into a more thermodynamically favorable crystalline state, then an amorphous solid will be formed. Because of entropy considerations, many polymers can be made amorphous solids by cooling even at slow rates. In contrast, if molecules have sufficient time to organize into a structure with two- or three-dimensional order, then a crystalline (or semi-crystalline) solid will be formed. Water is one example. Because of its small molecular size and ability to quickly rearrange, it cannot be made amorphous without resorting to specialized hyperquenching techniques.
Amorphous materials can also be produced by additives which interfere with the ability of the primary constituent to crystallize. For example, addition of soda to silicon dioxide results in window glass, and the addition of glycols to water results in a vitrified solid.
Some materials, such as metals, are difficult to prepare in an amorphous state. Unless a material has a high melting temperature ( e.g., ceramics) or a low crystallization energy (as polymers tend to), cooling must be done extremely rapidly. As the cooling is performed, the material changes from a supercooled liquid, with properties one would expect from a liquid state material, to a solid. The temperature at which this transition occurs is called the glass transition temperature or Tg.
Contents |
Definition
It is difficult to make a distinction between truly amorphous solids and crystalline solids if the size of the crystals is very small. Even amorphous materials have some short-range order at the atomic length scale due the nature of chemical bonding. Furthermore, in very small crystals a large fraction of the atoms are located at or near the surface of the crystal; relaxation of the surface and interfacial effects distort the atomic positions, decreasing the structural order. Even the most advanced structural characterization techniques, such as x-ray diffraction and transmission electron microscopy, have difficulty in distinguishing between amorphous and crystalline structures on these length scales.
The transition from the liquid state to the glass, at a temperature below the equilibrium melting point of the material, is called the glass transition. The glass transition temperature, Tg, is the temperature at which an amorphous solid, such as glass or a polymer, becomes brittle on cooling, or soft on heating. More specifically, it defines a pseudo second order phase transition in which a supercooled melt yields, on cooling, a glassy structure and properties similar to those of crystalline materials e.g. of an isotropic solid material.[1] Tg is usually applicable to wholly or partially amorphous solids such as common glasses and plastics (organic polymers). Below the glass transition temperature, Tg, amorphous solids are in a glassy state[2] and most of their joining bonds are intact. In inorganic glasses, with increased temperature more and more joining bonds are broken by thermal fluctuations so that broken bonds (termed configurons) begin to form clusters. Above Tg these clusters become macroscopic large facilitating the flow of material. In organic polymers, secondary, non-covalent bonds between the polymer chains become weak above Tg. Above Tg glasses and organic polymers become soft and capable of plastic deformation without fracture. This behavior is one of the things which make most plastics useful.[3] It is important to note that the glass transition temperature is a kinetic parameter, and thus parametrically depends on the melt cooling rate. Thus the slower the melt cooling rate, the lower Tg. In addition, Tg depends on the measurement conditions, which are not universally defined.[4] The glass transition temperature is approximately the temperature at which the viscosity of the liquid exceeds a certain value (about 1012 Pa·s). The transition temperature depends on cooling rate, with the glass transition occurring at higher temperatures for faster cooling rates. The precise nature of the glass transition is the subject of ongoing research. While it is clear that the glass transition is not a first-order thermodynamic transition (such as melting), there is debate as to whether it is a higher-order transition such percolation type transformation,[5] or merely a kinetic effect.
Amorphous thin films
Amorphous phases are important constituents of thin films, which are solid layers of a few nm to some tens of µm thickness deposited upon an underlying substrate. So-called structure zone models were developed to describe the microstructure and ceramics of thin films as a function of the homologeous temperature Th that is the ratio of deposition temperature over melting temperature.[6][7] According to these models, a necessary (but not sufficient) condition for the occurrence of amorphous phases is that Th has to be smaller than 0.3, that is the deposition temperature must be below 30% of the melting temperature. For higher values, the surface diffusion of deposited atomic species would allow for the formation of crystallites with long range atomic order.
Regarding their applications, amorphous metallic layers played an important role in the discussion of a suspected superconductivity in amorphous metals.[8] Today, optical coatings made from TiO2, SiO2, Ta2O5 etc. and combinations of them in most cases consist of amorphous phases of these compounds. Much research is carried out into thin amorphous films as a gas separating membrane layer.[9] The technologically most important thin amorphous film is probably represented by few nm thin SiO2 layers serving as isolator above the conducting channel of a metal-oxide semiconductor field-effect transistor MOSFET. Also, hydrogenated amorphous silicon, a-Si:H in short, is of technical significance for thin film solar cells. In case of a-Si:H the missing long-range order between silicon atoms is partly induced by the presence by hydrogen in the percent range.
The occurrence of amorphous phases turned out as a phenomenon of particular interest for studying thin film growth. Remarkably, the growth of polycrystalline films is often preceded by an initial amorphous layer, the thickness of which may amount to only a few nm. The most investigated example is represented by thin multicrystalline silicon films, where such an initial amorphous layer was observed in many studies, see for instance.[10] Wedge-shaped polycrystals were identified by transmission electron microscopy to grow out of the amorphous phase only after the latter has exceeded a certain thickness, the precise value of which depends on deposition temperature, background pressure and various other process parameters. The phenomenon has been interpreted in the framework of Ostwald's rule of stages[11] that predicts the formation of phases to proceed with increasing condensation time towards increasing stability.[8][10] Experimental studies of the phenomenon require a clearly defined state of the substrate surface and its contaminant density etc., upon which the thin film is deposited.
References
- ↑ The IUPAC Compendium of Chemical Terminology, 66, 583 (1997).
- ↑ A. Varshneya. Fundamentals of inorganic glasses. Boston, Academic Press (1994).
- ↑ But such behavior is not exhibited by crosslinked thermosetting plastics which, once cured, are set for life and will shatter rather than deform, never becoming plastic again when heated, nor melting
- ↑ O. V. Mazurin, Yu. V. Gankin: "Glass transition temperature: problems of measurements and analysis of the existing data"; Proceedings, International Congress on Glass, July 1-6, 2007, Strasbourg, France
- ↑ M.I. Ojovan. Glass formation in amorphous SiO2 as a percolation phase transition in a system of network defects. J. Exp. Theor. Phys. Let., 79 (12) 632-634 (2004)
- ↑ B. A. Movchan and A. V. Demchishin (1969). "Study of the structure and properties of thick vacuum condensates of nickel, titanium, tungsten, aluminium oxide and zirconium dioxide". Phys. Met. Metallogr. 28: 83–90.
- ↑ J.A. Thornton (1974). "Influence of apparatus geometry and deposition conditions on the structure and topography of thick sputtered coatings". J. Vac. Sci. Tech. 11: 666–670. doi:10.1116/1.1312732.
- ↑ 8.0 8.1 Buckel, W. (1961). "The influence of crystal bonds on film growth". Elektrische en Magnetische Eigenschappen van dunne Metallaagies. Leuven, Belgium.
- ↑ R.M. de Vos, H. Verweij (1998). "High-Selectivity, High-Flux Silica Membranes for Gas Separation". Science 279 (5357): 1710. doi:10.1126/science.279.5357.1710. PMID 9497287.
- ↑ 10.0 10.1 M. Birkholz, B. Selle, W. Fuhs, S. Christiansen, H. P. Strunk, and R. Reich (2001). "Amorphous-crystalline phase transition during the growth of thin films: the case of microcrystalline silicon". Phys. Rev. B 64: 085402. doi:10.1103/PhysRevB.64.085402.
- ↑ W. Ostwald (1897). "Studien über die Umwandlung fester Körper". Z. Phys. Chem. 22: 289–330.